Plastic and environment – do we mean the same or do we need a (standardised) glossary?

stics and environmental aspects – is there a need for a uniform terminology? Swedish experts at SIS have now created a glossary for this.
Do the postponements solve the problems with MDR?

The new MDR should provide better patient safety. New deferrals from the EU help in the transition but do not fix the structural problems with the MDR, which lead to subdued investment willingness within the EU.
10 tips for the industrialization of your plastic product

Do you have an idea that needs plastic parts? Take a few minutes to learn more about planning and realization of your idea for a successful market introduction.
Help! How do I give my medical device a CE mark?

VDE presents the process of CE road mapping in 10 steps.
Tips to become successful on the medtech market

A good product idea alone isn’t a guarantee for a successful market penetration.
The history of hemodialysis and the significance of plastics for dialysis

A brief history of hemodialysis and of how important plastics have been for its development.
How the new EU-MDR affects innovations and niche products

The idea behind EU’s new regulations for medical devices (MDR) was to secure the products’ safety and performance and thereby increase the patient safety. At the same time, MDR would enable further innovations. There are many indications that it became just the opposite.
How are plastics used in medical technology?
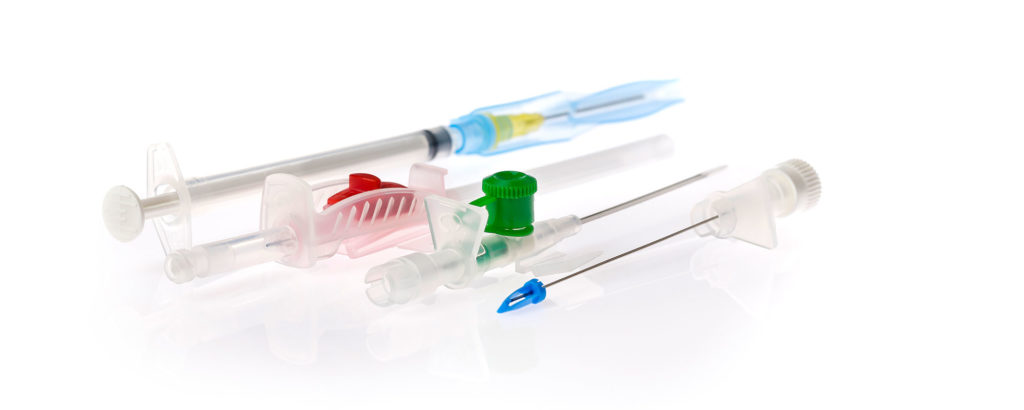
Plastic can not be thought out of our everyday lives, and this is especially true when we get sick. The material plastic has revolutionized the treatment possibilities and patient safety in the field of medicine, and many life-saving measures would not be possible at all without plastic. Here you get an overview of the importance of plastic in the medical field.
Financing in the medical technology industry – what opportunities are there?

Lack of venture capital can stop innovations. Various financing options from the German market that are also applicable in other countries.
The MDR is here – but what is the situation in the medtech industry?

Nya MDR gäller nu efter ett års uppskov men det finns fortfarande en del problemområden och oklarheter. Här ges en statusbeskrivning.