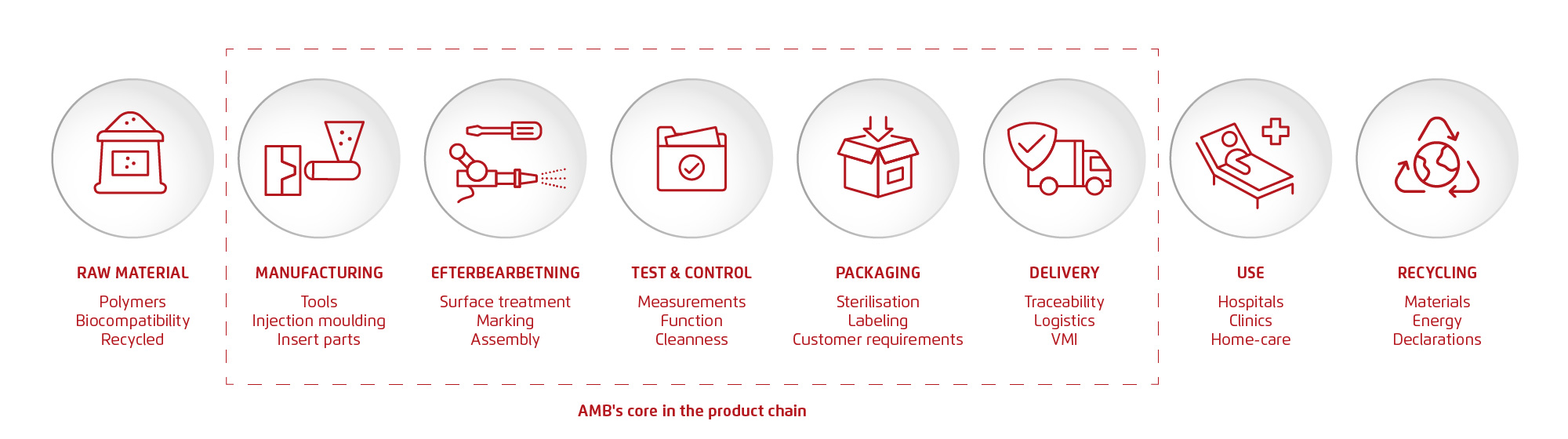
Life science is one of Sweden’s strongest growth industries, and medical technology accounts for the largest share of companies in the sector. As exports increase and the demands for quality, sustainability, and industrialization tighten, suppliers are becoming more important than ever. Here, we summarize the developments—and what they mean for AMB and our customers.
Introduction: How trends in Swedish Life Science affect manufacturing companies like AMB
Sweden’s life science sector is undergoing a period of significant transformation. Over the past decade, the field has grown in both economic importance and strategic relevance. It is no longer solely about pharmaceuticals and university hospitals—it is a complex ecosystem in which medical technology (hereafter referred to as medtech), advanced manufacturing, digitalization, and research form an interconnected value chain. At the center of this ecosystem are hundreds of manufacturing companies like AMB Industri AB, acting as hubs that connect technology, customer needs, quality systems, and industrial innovation.
Since 2021, the Swedish innovation agency Vinnova has continuously analyzed the sector’s development on behalf of the government. This article summarizes the key insights from Vinnova’s report “Life Science 2025” and explains how these developments affect the supplier landscape, particularly within medtech and the production of advanced plastic components. The focus is on practical implications: What do these trends mean for customers? And for a company like AMB, which today delivers advanced components, complete systems, and production solutions to medtech actors in Sweden and internationally?
Sweden is a strong Life Science cluster – and Medtech is the engine
The industry organization Sweden Bio divides life science into pharma, medtech, healthtech, diagnostics, biotech, and labtech. AMB operates primarily within medtech but also serves customers in healthtech and labtech. Within the life science sector, medtech is the largest segment in terms of number of companies; more than half of all businesses in the sector work with medtech in some form—from implants and disposable products to instruments, digital systems, and advanced production tools.
For a contract manufacturer like AMB, this is crucial. The medtech industry demands:
- components of exceptionally high quality
- repeatable and controlled production
- reliable suppliers
- traceability, documentation, and robust quality systems (such as ISO 13485)
- close collaboration between development and manufacturing
The growing number of medtech companies, especially small and medium-sized innovative actors, is driving demand for exactly the kind of technical and manufacturing expertise that AMB provides.
Six areas within Life Science
| Area | Definition |
| Medical Technology (Medtech) | Medical equipment and technology for diagnosis, treatment, and care—such as devices, instruments, implants, and surgical or therapeutic equipment, as well as e-health systems. Regulated under the EU Medical Device Regulation (MDR). |
| Pharmaceuticals | Chemically or biologically based medicinal products (medicines, vaccines, etc.). |
| Biotechnology | Development of biological drugs, therapies, cell and gene therapies, biomaterials, and regenerative medicine. |
| Diagnostics | Diagnostic tests (such as blood and tissue tests), analyses, and various types of diagnostic equipment. |
| Laboratory Technology | Laboratory instruments, analytical equipment, and services for laboratory work, diagnostics, and biotechnological research. This area often overlaps with medtech and biotech. |
Swedish exports are growing
One of the report’s clearest conclusions is that Swedish life science is heavily export-dependent. The export value of life science products has more than doubled over the past decade and now accounts for a full 10 percent of Sweden’s total exports. In 2023, Swedish life science companies exported goods worth SEK 196 billion. For comparison, the entire Swedish music industry generated SEK 6 billion in 2024. When will we start talking about “the Swedish life science miracle” in the same way that we talk about our proud music industry?
For a company like AMB, this development highlights two important points:
1. Customers operate in a global market
AMB’s customers in life science launch products in the USA, EU, Japan, and other regulated markets. The supplier chain must therefore meet the requirements of these markets in terms of quality, safety, and documentation.
2. Suppliers become strategic partners, not just manufacturers
As export markets grow more competitive, life science companies increasingly seek suppliers who can support:
- rapid industrialization
- stable high-volume production
- cost-efficient manufacturing
- materials expertise
- regulatory support
- development partnerships
AMB’s own R&D, testing capabilities, and experience in risk management therefore become key competitive advantages.
Research and education: Sweden remains strong – but challenges lie ahead
The report shows that Sweden ranks highly internationally in terms of the number of researchers, doctoral students, and scientific publications per capita. Uppsala, Stockholm, Gothenburg, Lund, and Umeå form strong research hubs and drive much of the innovation capacity within Swedish life science.
At the same time, a growing competence gap is emerging. Fewer engineers are graduating in several critical technical fields, and production-related expertise is becoming increasingly difficult to recruit. Competition for specialists in quality, RA/QA, and industrialization is also intensifying—not least because global corporations attract skilled experts away from the Swedish market.
In this landscape, companies like AMB play an important role. By offering attractive and long-term career opportunities in areas such as production engineering, material development, project management, validation, and quality assurance, AMB helps strengthen the talent pipeline across the entire Swedish life science sector.
Distribution of Swedish Life Science companies by region
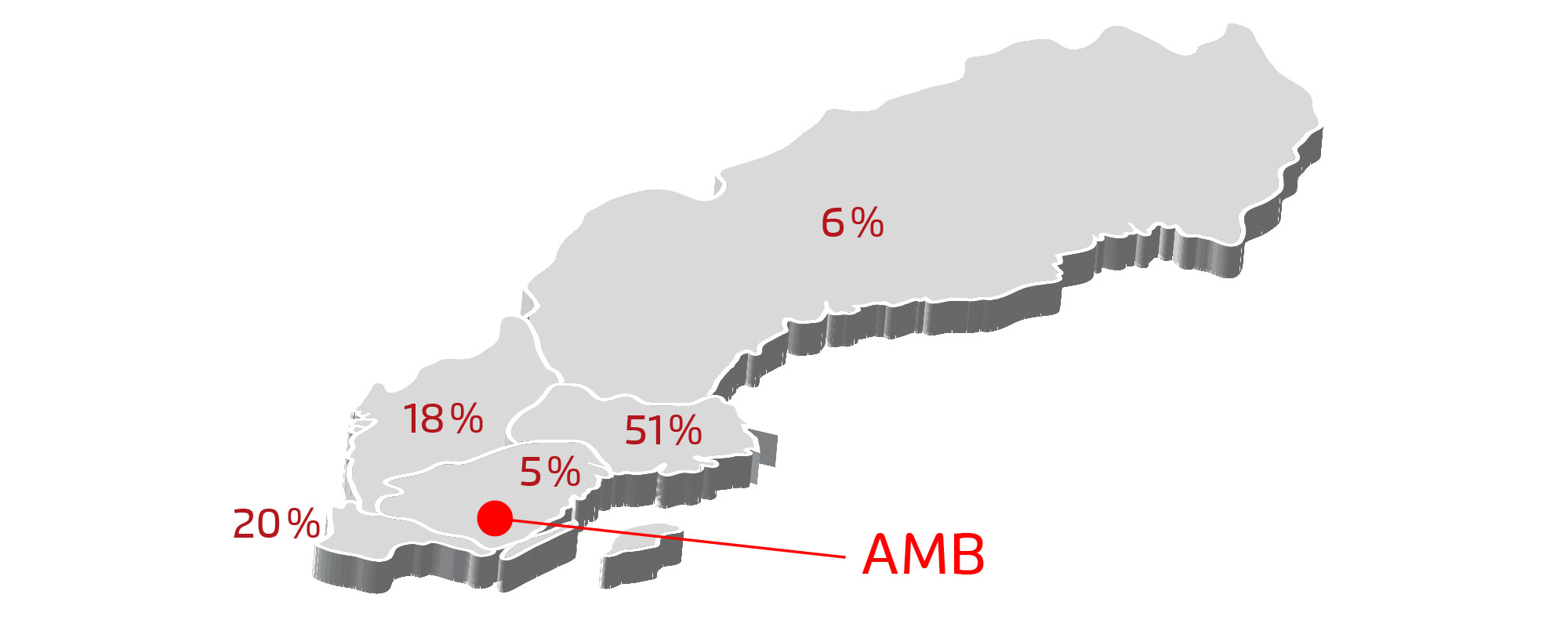
Strong growth in the number of companies – but major differences in maturity
Sweden’s life science sector has grown rapidly in the number of companies, particularly among small and medium-sized medtech firms. The largest concentrations are found in:
- Västra Götaland
- Stockholm
- Skåne
- Uppsala
Many of these companies are technology-driven, research-oriented and highly innovative. But as they scale up, a common challenge emerges: they often lack in-house manufacturing capacity. Building and validating their own production facilities is costly, highly regulated and capital-intensive.
This is driving a growing demand for partner companies like AMB, which already offer:
- qualified production in a wide range of polymer materials
- validated and controlled processes
- cleanroom manufacturing
- extensive experience with complex medical device components
- close, long-term customer collaborations
Industry shift: From production to innovation – and back again
A clear trend is that Swedish life science has historically shifted from manufacturing toward research and design. But in recent years, the pendulum has begun to swing back. There is now a growing understanding that production is a strategic asset—especially in turbulent times marked by global uncertainty, supply-chain disruptions, and geopolitical risks.
The report emphasizes the need to “strengthen Sweden’s life science manufacturing capabilities.” This aligns closely with what AMB has observed in customer interactions:
- More Swedish companies want suppliers located nearby
- Quality and delivery reliability outweigh price
- Collaboration around industrialization is becoming more important than pure manufacturing
- Sustainability and traceability are gaining significance
- Prototype-to-production timelines must be shorter
With decades of experience in injection molding, surface treatment, and industrialization, AMB is well positioned to act as an extended workshop, development partner, and production advisor for life science customers with high demands and high ambitions.
Sustainability: From requirement to competitive advantage
Sustainability is no longer a side issue—it is a central part of the development of the life science sector.
The report highlights in particular:
- requirements for material selection
- traceability
- energy use
- production methods
- transport efficiency
- waste management
- circular flows in packaging and single-use products
For us at AMB, this means that our work with material development, process optimisation and energy-efficient production is not just internal improvement work—it is a service to the customer.
Life science companies often need:
- documented material choices
- the option to use recycled raw materials where possible
- reduced weight through improved design
- fewer process steps
- energy-efficient production methods
Through our own development expertise and close collaboration with material suppliers, AMB can support customers in meeting their sustainability goals—helping them comply with regulatory requirements and the growing expectations for sustainability reporting (ESG).
Subcontractors as strategic assets—not costs
In the report, the Swedish life science sector is described as increasingly dependent on specialized suppliers rather than fully vertically integrated companies. This is especially true in the medtech segment, where product development and commercial strategies are often driven by relatively small teams.
- For AMB, this means that customers need far more than production capacity alone. They need:
- Industrialization support
- Material expertise
- DFM (Design for Manufacturability)
- Early-stage risk assessments
- Quality-assured pilot runs
- Robust scalability for volume production
- Documentation aligned with regulatory requirements
The practical reality in the industry is clear: Customers increasingly want fewer—but stronger—suppliers, and the partnerships are becoming more long-term and strategic.
The future: Three trends where AMB plays an important role
1. Increased specialization in component manufacturing
Medical technology products are becoming increasingly complex—in materials, geometries, and tolerances. Plastic components often require:
- high dimensional stability
- biocompatibility
- integration with electronics, software, or sensors
- cleanroom manufacturing
- advanced testing requirements
- surface treatments (such as EMC shielding or cosmetic coatings)
AMB’s investments in technology, production equipment, and in-house expertise directly match these needs. We also have extensive experience in the logistics required for high-volume production involving many part numbers, including full VMI solutions where we manage customers’ inventory levels.
2. Stronger integration between development and production
Innovation in medtech is accelerating. Customers need a faster path from idea to finished product. Suppliers with their own development expertise and validated processes can shorten lead times and reduce risks.
3. Sustainability requirements driving material innovation
Plastics remain dominant in medtech. Growing sustainability demands are pushing for:
- lighter designs
- reduced waste
- better material choices
- energy-efficient production
AMB’s deep knowledge of polymer materials and production optimization helps customers meet their ESG targets.
Summary: Life Science is growing – and the supplier chain must grow with it
The Swedish life science sector is stronger than it has been in decades. Exports are rising, research output is growing, and the number of companies is higher than ever. But with growth comes increased demands—especially around quality, sustainability, industrialisation, and delivery reliability.
For companies like AMB, this brings significant opportunities:
- More life science actors are seeking long-term, reliable partners
- European production is once again becoming a key strategic asset
- Sustainability and uncompromising quality are no longer competitive advantages—they are basic expectations
- Global expansion among customers requires robust manufacturing and a supplier capable of scaling alongside them
As a supplier within medtech, healthtech, and labtech, AMB is positioned at the centre of this development—and is ready to continue supporting both Swedish and international life science companies as the industry grows.